Extracellular Matrix-based Therapy Development Timeline
The Badylak lab develops the first bioscaffold for use as a surgical mesh applications
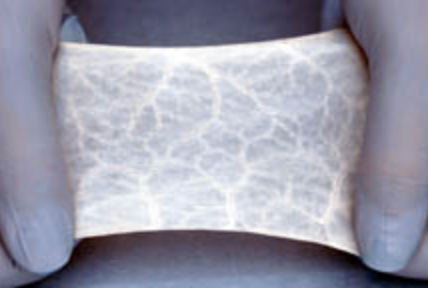
FDA approval of the first ECM-based bioscaffold.

Tunable ECM hydrogels from decellularized ECM were discovered by the Badylak Laboratory at University of Pittsburgh

on ECM-based therapies
The Badylak laboratory discovered Matrix-Bound Nanovesicles (MBV) embedded within ECM Bioscaffolds.
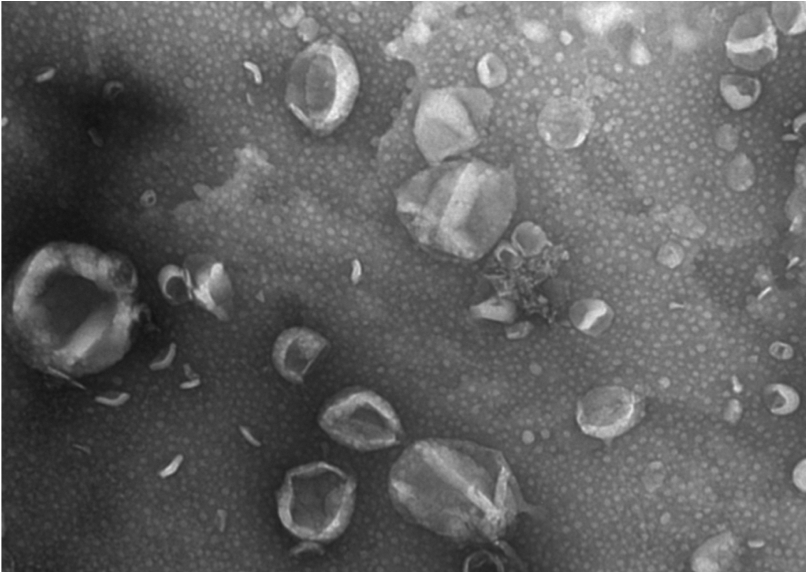
Launch of ECM Therapeutics to commercialize injectable ECM Hydrogel and Matrix-Bound Nanovesicles (MBV) therapies

Submission of 510(k) application for ECM hydrogel for wound management.

Initiation of IDE clinical study evaluating injectable ECM hydrogel for anorectal fistula repair.




